India has one of the lowest testing rates for Covid-19 in the world at 0.04 tests per 10K population
MyLab gained renown by becoming the first startup to develop an indigenous testing kit
Redcliffe has partnered with BSIP Lucknow to conduct tests using its expertise
The Crusaders
Latest innovations, jugaad, and initiatives taken by India's tech ecosystem & leaders to help India flatten the curve and navigate through the Covid-19 impact.
Last month, the story of Minal Dakhave Bhosale was reported extensively as India’s attention was fixed on coronavirus testing. Bhosale, head of the research and development team at Pune-based biotech startup MyLab, had achieved the feat of developing Covid-19 testing kit within six weeks, just a day before giving birth.
The indigenous Covid-19 kit developed by MyLab had come at a time when India was desperately looking to tackle the low testing rate for the virus. At the time, India was on the verge of falling behind the required rate of testing to catch cases early, before they spread. But thanks to various interventions, including MyLab’s testing kit, India managed to reign in the spread much better than countries like the US and Italy.
However, experts suggest that India is still testing a lot less than those countries, due to which the number of confirmed positive coronavirus cases in India has remained quite low.
According to the official data, India is testing approximately 0.04 individuals for every 10,000 people, as of April 9. The number is quite low as compared to Spain and Italy which are testing around 18.3 and 15.8 individuals per 10K population.
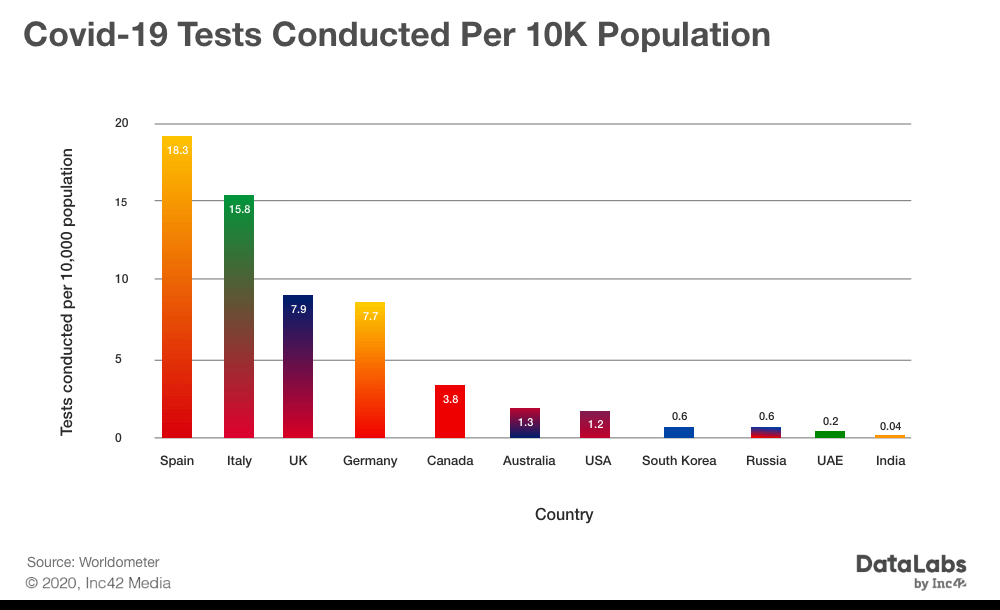
On the other hand, the Indian government is claiming that appropriate numbers of testing have been conducted. Notably, over 350K tests have been conducted until April 17. But with the number of cases expected to rise sharply in the future, many have started to question whether the government has an appropriate number of testing kits or not.
So is India armed well for the long battle? According to some biotech startups, India has controlled the situation well till now, but there will be a need for further ramping up of testing and that’s where most of their efforts are centred around.
As of now, only a handful of startups have developed their own Covid-19 testing kits. These include MyLab, Redcliffe Life Sciences, Genes2Me, CoSara Diagnostics, FastSense Diagnostics, and Bione. However, the title for developing the first practically usable kit in India will always remain with MyLab.
Gururgam-based Genes2Me has successfully delivered 2.5 Lakh rapid test kits to the government in a record time of seven days while CoSara Diagnostics is aiming to provide around 10K per day as its contribution to the battle against coronavirus.
Meanwhile, Pune-based FastSense Diagnostics is said to be developing two new ways to diagnose Covid-19. On the other hand, Bione had recently launched a rapid Covid-19 at-home screening test kit. Bione said that it was the first healthcare company in India to achieve the feat.
The Story Of India’s First Covid-19 Testing Kit

Recalling the development process for the testing kit, MyLab said that it just took six weeks for the company to launch the product. The research and development team at MyLabs started working on the kit in February, much before the situation worsened in India. “Looking at how Covid-19 was spreading across the world, our research team started working on developing the test,” MyLab said.
Founded in 2016, MyLab has created a reverse transcription-polymerase chain reaction (RT-PCR) kit — Mylab PathoDetect COVID-19 Qualitative PCR — which is manufactured domestically, has a local supply chain and is approved by Indian Council of Medical Research (ICMR).
The kit developed by the company is said to be a quarter of the cost of international counterparts. MyLab also claims that the screening can be done within 2.5 hours which is much lower than the seven or eight hours taken by the kits procured by the government from other countries.
As of now, the company is making enough kits for around 1.5 Lakh tests per week. But given the spike in the number of cases, MyLab is ramping up the manufacturing capabilities to test around 20 Lakh samples in a week.
To achieve this, MyLab said that multiple investors have shown their interest in the company while a few have already infused the funds. MyLab has also secured INR 1 Cr from the Action Covid-19 Team (ACT) grants with the group of founders, investors, and venture capitals also helping the company solve supply-chain issues.
Also, Serum India CEO Adar Poonawalla and AP Globale Chairman Abhijit Pawar have backed the company to scale the manufacturing of test kits. MyLab said that it is automating the plant production and processes with the recently received grants and funds.
Currently, MyLab is supplying diagnostic kits to many state governments in India. For example, the Chhattisgarh government has procured more than 1000 test kits from MyLab while Chandigarh and others are also placing their orders with the company.
Besides being the first company to develop the Covid-19 test kits, MyLab also claims to be the only Indian biotech player to have approval for conducting individual donor nucleic acid testing. The test is used to screen donor blood samples which can detect potential risks of transfusion-transmitted infections in recipients.
Healthcare Gets A Helping Hand From Tech

After MyLab’s discovery, the race between Indian biotech startups to create more testing kits intensified. As a result, many startups came up with unique propositions, including New Delhi-based Redcliffe Life Sciences.
Founded by Dheeraj Jain and Ashish Dubey in 2018, Redcliffe is molecular genomics, precision medicine and research startup that offers genomics testing support for mother and child. It is now working solely on Covid-19 diagnosis kits.
The story started with Dr Niraj Rai, who runs the ancient-DNA lab at Lucknow-based Birla Sahni Institute of Palaeosciences (BSIP), calling Dubey. “During the call, Rai said that he wanted to help the situations but doesn’t have enough resources to do so,” Dubey told Inc42.
On the other hand, Redcliffe had the expertise as scientists working for the startup had experience in a similar domain. So Dubey was quick to propose a partnership with Redcliffe which happened in no time. Dubey said, “I asked four of my team members to help BSIP and they agreed to help. All of them are now in Lucknow.”
Dubey said that BSIP had the biosafety lab (BSL Lab). Seeking this opportunity, Redcliffe struck a memorandum of understanding (MoU) with BSIP. “Now we are providing the expertise and BSIP is providing the infrastructure,” Dubey added.
After this, the startup went for approvals and got them in no time. Dubey said Redcliffe got approval last week from the department of science and technology while the UP health ministry nodded its proposition earlier this week.
As a result, a PPP model has been devised with the government within two weeks to conduct RT-PCR testing in Uttar Pradesh. Dubey said by Monday, the testing will start at BSIP labs. “We are doing our internal setup at BSIP before the commercialisation of these tests from Monday,” he added.
As far as the pricing is concerned, Redcliffe will be charging INR 4500 for private samples while all the samples sent by the government hospitals will be tested at INR 3500. Dubey said that all the tests conducted under the Ayushman Bharat Yojana will be free now and the reimbursement will be later discussed with the government.
Besides the RT-PCR testing, Redcliffe is working on a mass-screening assay to diagnose a large population with the help of Dr Rai’s team at BSIP. Dubey said that the mass-screening assay can diagnose around 3000 samples at one go. Also, the test would be cheaper as compared to the current prices of RT-PCR tests. “The cost of these tests would come around INR 700,” Dubey noted.





























 Ad-lite browsing experience
Ad-lite browsing experience